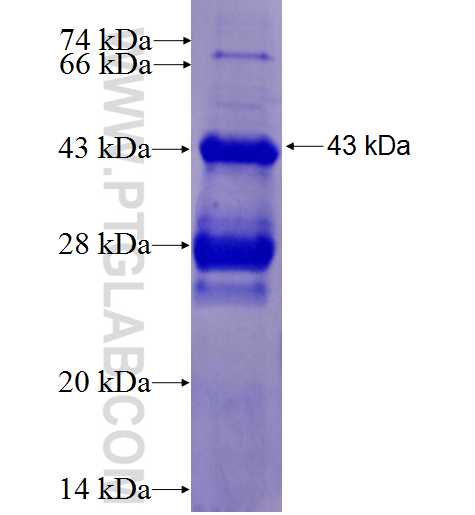
Activation of Saccharomyces cerevisiae Mlh1-Pms1 Endonuclease in a Reconstituted Mismatch Repair System* - Journal of Biological Chemistry

PCNA and Msh2-Msh6 activate an Mlh1-Pms1 endonuclease pathway required for Exo1-independent mismatch repair. | Semantic Scholar

PMS1 Mouse anti-Human, Clone: OTI5E10, TrueMAB™, Origene 30 μL; Unconjugated Products | Fisher Scientific

Structure of the MutLα C-terminal domain reveals how Mlh1 contributes to Pms1 endonuclease site | Nature Structural & Molecular Biology

Identification of MLH2/hPMS1 dominant mutations that prevent DNA mismatch repair function | Communications Biology

The unstructured linker of Mlh1 contains a motif required for endonuclease function which is mutated in cancers | PNAS






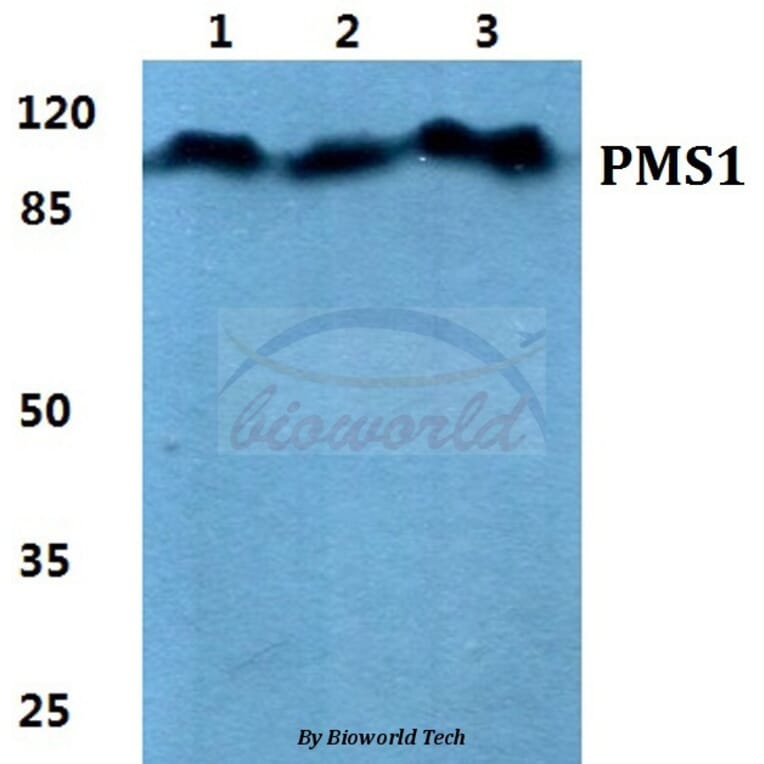
![Anti-PMS1 Antibody [PCRP-PMS1-2E11] (A278340) | Antibodies.com Anti-PMS1 Antibody [PCRP-PMS1-2E11] (A278340) | Antibodies.com](https://cdn.antibodies.com/image/catalog/278/A278340_2.jpg?profile=product_top)


-Western-Blot-NBP2-09011-img0001.jpg)